Iron-oxides are usually formed by precipitation from iron-enriched groundwater. Iron-oxide pigments consist of ferrous or ferric oxides, and impurities such as clay and/or manganese. Natural iron-oxide pigments have been used for centuries as coloring agents. Iron-oxide pigments are valued because they are nontoxic, weather resistant and they do not bleed or fade. The pigments are extracted from a range of ores including hematite, goethite, limonite, siderite, and magnetite. Hematite produces red pigments, limonite and goethite produce yellow and brown pigments, siderite produces brown and red pigments, and magnetite yields black pigments.
As of 2023 , only two states actively mined iron oxide pigments, Alabama and Georgia (USGS, 2024). Until 2013, iron-oxide mining had occurred in Virginia at two locations (Hiwassee, Pulaski County and Painter Mine, Wythe County). Hoover Color Corporation and its predecessors produced pigments from local materials between 1912 and 2013. Pigments included ocher, sienna, and umber for a variety of products including paint, varnishes, fertilizer, cement, clothing, inks, crayons, cosmetics, and linoleum tile (Figure 55, Southwest Times, 1955; Johnson, 1964; Paine, 2024). Open pit methods were used to mine these natural iron-oxides. The iron-oxides were formed by precipitation from iron-enriched groundwater, near the contact of the Cambrian (543 to 490 million years ago) Erwin Formation with the overlying Cambrian Shady Dolomite. Deposits, associated with gossans formed from weathering of the Cambrian rocks, are concentrated as small bodies or pockets composed of insoluble clay and iron oxide.
In 2003, approximately 519 tons of natural iron-oxide were reportedly mined and produced from stockpiles in Virginia, making it the fourth largest producer in the United States (Potter, 2003). The highest production of the 21st century occurred in 2000, with approximately 821 short tons reported from the two separate operations in Pulaski and Wythe County. After reclamation was completed in the late 2010s, the Hiwassee location was acquired by the Commonwealth for inclusion into the New River Trail State Park and was open to the public in 2019. More details on historical operations at the Hiwassee mine can be found in the 2024 Virginia Geological Field Conference guidebook (VGFC, 2024).
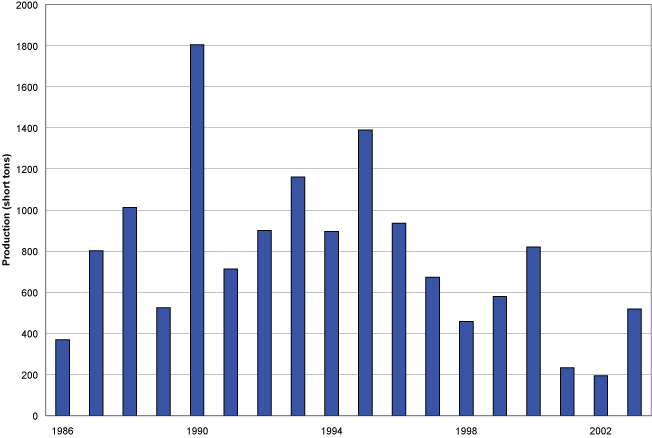
Iron-oxide production in Virginia, 1986-2003.
Selected References:
Gilmer, A. K., Enomoto, C. B., Lovett, J. A., and Spears, D. B., 2005, Mineral and fossil fuel production in Virginia (1999-2003): Virginia Division of Mineral Resources Open-File Report 05-04, 77 p.
>Heller, M.J., Watts, C.F., Henika, W.S., Orndorff, W.D., McClellan, E.A., Swanger, W.R., Prince, P.S., and Tso, J.L., 2024, Virginia Geological Field Conference, November 1-3, 2024, Field Guidebook. Available upon request.
Johnson, S.S., 1964, Iron and titanium mineral pigments in Virginia: Virginia Minerals, v. 10, n. 3, p. 1-6.
Paine, W., 2024, “Hoover Color closes after vivid history,” Southwest Times: Patriot Publishing, Pulaski, VA, 4 June 2024.
Potter, Michael J., 2003, Iron-Oxide Pigments: U.S. Geological Survey Minerals Yearbook, p. 41.1-41.3.
Southwest Times, 1955, “American Pigment operates two plants in Pulaski County,” Southwest Times: Pulaski, VA, V. 49, N. 21, 16 January 1955.
USGS, 2024, National Minerals Information Center, Iron Oxide Pigments Statistics and Information .
Virginia Division of Mineral Resources, 1993, Geologic Map of Virginia: Virginia Division of Mineral Resources, scale 1:500,000.
