Management of greenhouse gas emissions has become a major issue for governments and the industries whose emissions they regulate. Concerns about global warming are driving policy decisions that may soon bring about major changes in the way greenhouse gases are managed in the United States. Carbon dioxide (CO2), a product of fossil fuel combustion, is one of the larger contributors to greenhouses gases.
A technology with the potential to mitigate the amount of CO2 released to the atmosphere as fossil fuel emissions is known as Carbon Dioxide Capture and Storage (CCS). This involves the capture of CO2 at point sources such as coal power plants, and injecting it directly into deep underground geological formations. Oil fields, gas fields, saline formations, unmineable coal seams, and saline-filled basalt formations have been suggested as possible storage sites (Figure 1).
Two important geologic factors that can act as trapping mechanisms to prevent leakage of injected CO2 back to the surface include:
- Depth of burial and the presence of low-permeability caprocks or stratigraphic seals;
- Geochemical reactions of CO2 with host rocks and the formation of stable minerals (Gunter and others, 2004).
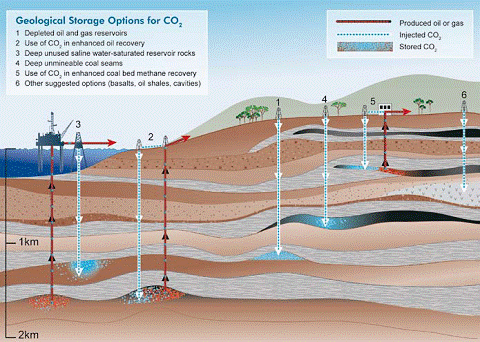
Figure 1: Options for storing CO2 in deep underground geological formations (after Cook, 1999)
Enhanced Oil and Gas Recovery
The injection of CO2 into deep geological formations has been on-going for over 40 years. It was first undertaken in Texas in the early 1970s as part of enhanced oil recovery (EOR) projects, and has been on-going there and at many other locations around the world ever since. In the U.S., approximately 30 to 50 million metric tons of CO2 are injected annually into oil fields that are declining in oil production (Benson and others, 2005). The CO2 used in the EOR projects is collected, in part, from anthropogenic sources, but is principally derived from naturally occurring geologic sources of CO2. It is transported to the oil-producing fields through a large network of pipelines.
The first large-scale CCS project was initiated in 1996 by the Norwegian company Statoil to remove excess CO2 from natural gas produced from deposits in the North Sea. At Sleipner, the company strips CO2 from the natural gas and stores it in a deep saline aquifer.
In 2000, a coal-fueled synthetic natural gas plant in Beulah, North Dakota became the world's first synfuel plant to capture and store carbon dioxide produced from coal conversion. Dakota Gasification Company captures about 3 million tons of CO2 annually at the Great Plains Synfuels Plant, and transfers it via a 205-mile pipeline to the Weyburn-Midale oil field located in southeast Saskatchewan, Canada. There the CO2 is used for enhanced oil recovery and permanently stored in the depleted oil reservoir. http://ptrc.ca/projects/weyburn-midale.
Assessing Geologic Reservoirs for CO2 Storage
In 2007 the U.S. Geological Survey (USGS) was authorized to conduct a national assessment of potential geologic storage resources for CO2 in cooperation with the U.S. Environmental Protection Agency and the U.S. Department of Energy under the Energy Independence and Security Act (Public Law 110–140). The USGS uses the following criteria for considering geologic formations as potential storage units:
- A minimum depth of 3,000 feet (914 m) below ground surface; CO2 at this depth is typically subjected to temperatures and pressures that maintain the CO2 in a supercritical state. Deep sequestration also helps insure there is an adequate thickness of rock (confining layers) above the potential injection zones to act as a geologic seal;
- Proposed salinity limit of at least 10,000 parts per million (ppm) for total dissolved solids (TDS) of formation waters;
- Geologic formations with the potential to store a minimum of 1 million metric tons (or greater) of CO2.
CO2 mineralization
An alternative to CO2 storage as a fluid in sedimentary reservoirs is a geochemical method, CO2 mineralization, which occurs via carbonation reactions. In this process, CO2 reacts with rocks and minerals to form solid and stable carbonate rocks. CO2 mineralization contains fewer long-term CO2-leakage concerns compared to saline storage methods and therefore potentially lower long-term monitoring costs. The suitable rock types for large-scale CO2 mineralization are rocks with high magnesium (Mg), calcium (Ca), or iron (Fe) content. Desirable rocks that are abundant in the United States include the ultramafic rocks, dunite, peridotite, serpentinite, and the mafic rock basalt (Blondes and others, 2019).
Current Work:
Virginia Energy is collaborating with Virginia Tech and conducting a carbon mineralization and metal extraction (CMME) study. This project is funded by Mining Innovations for Negative Emissions Resource Recovery (MINER) Program. The project team is aiming to develop an innovative CMME technology that enables the recovery of energy-relevant elements during direct and indirect carbon mineralization processes. The CMME technology will be tested on low-grade mafic and ultramafic rocks and allanite ore, which could supply huge resources of nickel, cobalt, and rare earth elements. Geologists from Virginia Energy will coordinate the collection of samples from mafic and ultramafic rocks formations to provide feedstocks for CMME testing (Figure 2). Our sampling effort will focus on 1) the mafic-ultramafic geologic units of Proterozoic age, including the meta-basalt within the Catoctin Formation, the soapstone at Albemarle-Nelson Soapstone Belt, and other ultramafic (komatiite) units identified from geologic maps and published literature; and 2) the mafic diabase rocks of Jurassic age. Based on the CMME test results, Virginia Energy will perform a preliminary resource assessment of energy-relevant elements in mafic and ultramafic rocks in the study area.
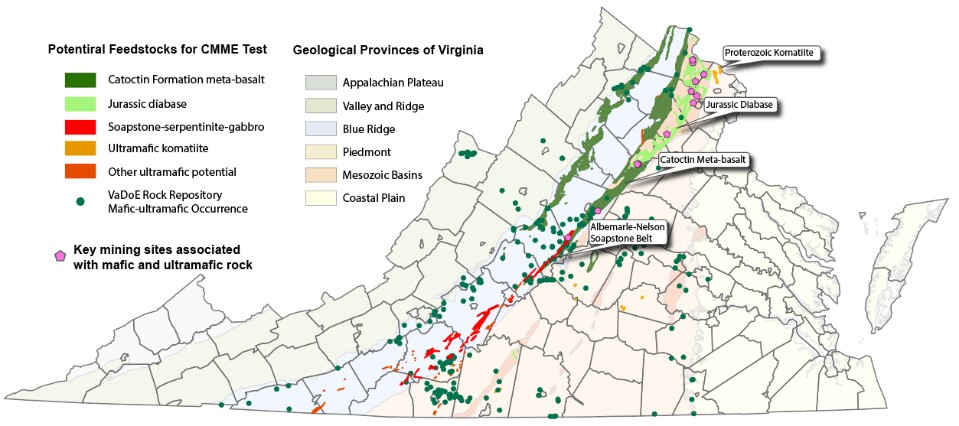
Figure 2: Map showing potential carbon mineralization and metal extraction resources in Virginia
Past CCS Projects:
In 2010-11, Virginia Energy in cooperation with the USGS conducted a preliminary assessment of the deep geologic formations in Virginia that might serve as permanent storage formations for CO2 (Figure 3). This work was supported by a grant from the United States Geological Survey (USGS) as part of the National Geologic Carbon Dioxide Sequestration Assessment. The storage formations and potential confining caprocks were identified and their structure, depth, and thickness were mapped. Physical data pertinent to CO2 storage, such as porosity, formation salinity, and permeability were compiled where the information was available. A comprehensive series of digital maps and databases were constructed. An example of the type of map produced for this study is shown in Figure 4. This research has shown that Virginia has the storage resources available for permanently storing CO2 in deep geological reservoirs, particularly in the gas fields of southwest Virginia. Future work should include further geologic mapping and modeling of additional stratigraphic intervals as well as the refinement of those maps and models developed during this phase of the project.
A PDF copy of the open-file report is available upon request.
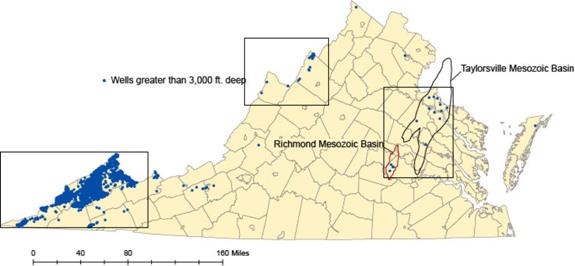
Figure 3: Areas of investigation (annotated by boxes) based on locations of wells completed at depths greater than 3,000 ft
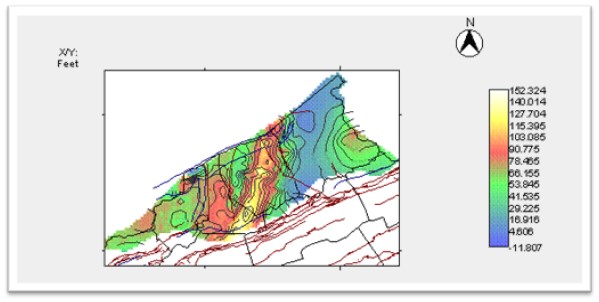
Figure 3: Thickness map of the Berea Sandstone at depths greater than 3,000 ft in southwest Virginia. The thickness of the sandstone greatly increases in the Nora and Break-Haysi Gas Fields in Dickenson and Buchanan Counties
In 2011, funded by Southern States Energy Board (SSEB), Virginia Energy and Virginia Tech conducted a preliminary investigation on the carbon sequestration potential within the Virginia Piedmont and Coastal Plain (Roth and others, 2012; Figure 5). This research evaluated the feasibility of CO2 storage into deep saline aquifers of the Mesozoic-age sedimentary basins and Atlantic Coastal Plain. Based on the regional investigation, multiple buried and exposed Mesozoic basins and Potomac Formation aquifer in offshore Virginia are suitable for large-scale storage of CO2. These potential reservoirs are located in close proximity to the CO2 emission sources for developing the capture and storage infrastructure. The estimated CO2 storage capacities for potential storage units range from 0.05 to 14 MMtonnes of CO2.
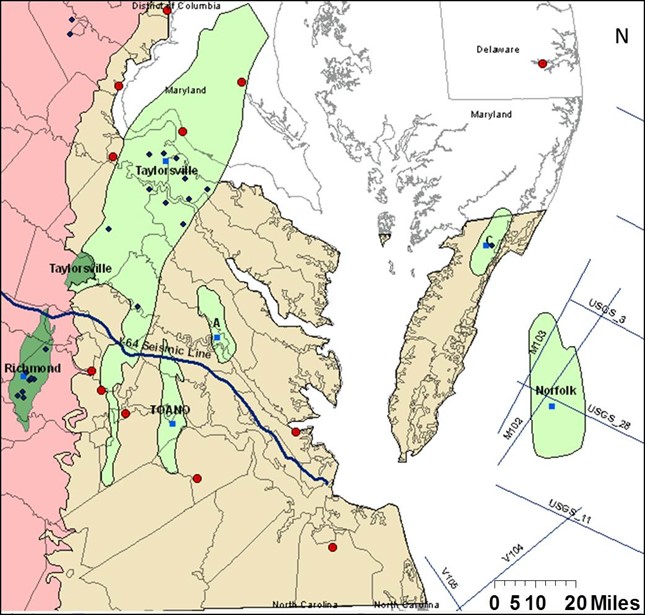
Figure 5: Map showing the inferred Mesozoic basins (light green) and the exposed basins (dark green) of the project. The red circles represent the significant sources of CO2 on the east coast, the blue squares show the proposed injection locations, and the dark blue diamonds represent oil and gas wells drilled to depths of 2,400ft and deeper in the Mesozoic basins of Virginia (Modified from Roth and others, 2012)
A PDF copy of the project publication is available in HERE.
In 2015-17, Virginia Energy worked cooperatively with Virginia Tech under a project funded by the U.S. Department of Energy (DOE). In this project, a total of 13,263 ton (12,032 Mt) of CO2 was injected into three coalbed methane wells in Buchanan County, Virginia. The primary objective of the research was to test the injectivity of CO2 into unmineable coal seams and the potential for enhanced coalbed methane recovery (ECBM). The results demonstrated success in characterizing the behavior of the coal reservoir in response to CO2 injection, thus providing data to better understand the effect of matrix swelling during injectivity and ECBM.
A PDF copy of the project publication is available in HERE.
In 2015-19, Virginia Energy conducted a DOE-funded project on Southeast Offshore Storage Resource Assessment partnered with Southern States Energy Board (SSEB). This project aimed to assess prospective geologic storage resources for CO2 in the State and Federal waters of three planning areas: the Mid-Atlantic, the South Atlantic, and the eastern Gulf of Mexico. Results of this research suggested that about 16 Gt of CO2 could be stored in the Cretaceous sandstone units offshore of Virginia beneath the continental shelf.
A PDF copy of the project publication is available in HERE.
Selected References:
Blondes, M.S., Merrill, M.D., Anderson, S.T., and DeVera, C.A., 2019, Carbon dioxide mineralization feasibility in the United States: U.S. Geological Survey Scientific Investigations Report 2018-5079, 29 p., https://pubs.er.usgs.gov/publication/sir20185079.
Cook, P.J., 1999, Sustainability and nonrenewable resources: Environmental Geosciences, 6(4), 185–190.
Gunter W.D., Bachu S., and Benson S., 2004, The role of hydrogeological and geochemical trapping in sedimentary basins for secure geological storage of carbon dioxide, in: Baines S.J., and Worden R.H.,( Eds.), Geological Storage of Carbon Dioxide: Geological Society of London, Special Publication 233, pp 129–145.
Karmis, Michael, Ripepi, Nino, Gilliland, Ellen, Louk, Andrew, Tang, Xu, Keles, Cigdem, Schlosser, Charles, Diminick, Ed, McClure, Michael, Hill, Gerald, and Hill, Brian. Central Appalachian Basin Unconventional (Coal/Organic Shale) Reservoir Small Scale CO2 Injection Test. United States: N. p., 2018. Web. doi:10.2172/1439921
Metz, B., Davidson, O., de Coninck, H., Loos, M., and Meyer, L. (Eds.), 2005, IPCC Special Report on Carbon Dioxide Capture and Storage: Intergovernmental Panel on Climate Change (IPCC), Cambridge University Press, U.K., 431p.
Nemeth, Ken, Sams-Gray, Kimberly, Berry, Patricia, Ripepi, Nino, Pashin, Jack, Knapp, James, Hills, Denise, and Riestenberg, David. Southeast Offshore Storage Resource Assessment (SOSRA) (Final Technical Report). United States: N. p., 2019. Web. doi:10.2172/1606208.
Roth, Ben, Hernon, Katie, Lassetter, William, and Nino Ripepi. "Evaluation of Large-Scale Geologic Carbon Sequestration Potential in the Virginia Piedmont and Coastal Plain." Paper presented at the Carbon Management Technology Conference, Orlando, Florida, USA, February 2012. doi: https://onepetro.org/CMTCONF/proceedings-abstract/12CMTC/All-12CMTC/CMTC-151252-MS/600
